What is diffusion and how does it apply to the process of chemiosmosis?
 A diagram of diffusion. As time passes, more of the particles diffuse into the side with a smaller concentration of those particles.
A diagram of diffusion. As time passes, more of the particles diffuse into the side with a smaller concentration of those particles.
Imagine you have a dam filled to the brim with water. On the other side of the dam, there is a very deep ditch. Since the water flow is blocked by a wall, the water cannot flow into the ditch. But what would happen if we created a hole in the dam? Water would inevitably start flowing through the hole and fill the ditch until there was an equal amount of water on both sides of the wall. This principle is called diffusion. Since there is a high concentration of water in the dam, the natural flow of the water would be into an area with a smaller concentration of water, like the ditch. The same can be said of the diffusion that occurs within the context of chemiosmosis. As the protons are pumped through protein channels into the intermembrane space or the thylakoid space, a buildup of protons occurs in those areas.
In addition to forming a greater hydrogen concentration gradient on one side of the membrane, the protons also form an electrical gradient due to the fact that they carry a positive charge (H+, or a proton, is basically a hydrogen atom with its one electron stripped away from it). As the diffusion process continues, more electrons and protons are fed through the system, and consequently more protons are pumped into the intermembrane or thylakoid space. This causes the hydrogen ions to become more inclined to traverse the membrane once again and evenly disperse throughout the intracellular space as well, something that is used in the production of ATP by ATP synthase.
In addition to forming a greater hydrogen concentration gradient on one side of the membrane, the protons also form an electrical gradient due to the fact that they carry a positive charge (H+, or a proton, is basically a hydrogen atom with its one electron stripped away from it). As the diffusion process continues, more electrons and protons are fed through the system, and consequently more protons are pumped into the intermembrane or thylakoid space. This causes the hydrogen ions to become more inclined to traverse the membrane once again and evenly disperse throughout the intracellular space as well, something that is used in the production of ATP by ATP synthase.
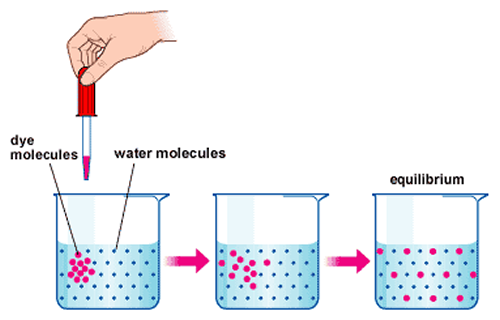 Dye molecules and water molecules at chemical equilibrium. Notice that the two types of particles are evenly dispersed throughout the beaker.
Dye molecules and water molecules at chemical equilibrium. Notice that the two types of particles are evenly dispersed throughout the beaker.
You may be wondering what would happen if a limited number of protons were allowed outside of the membrane. To answer this question, take a look at the picture on the left, illustrating the interaction of dye molecules with water molecules. A dense concentration of dye molecules is added to the beaker, and begins to even out in accordance with the principle of diffusion. Eventually, the two types of particles are evenly dispersed throughout the beaker- this is an example of the concept of equilibrium. Equilibrium occurs when a substance/substances is evenly divided in terms of concentration everywhere in the reaction chamber. In chemiosmosis, equilibrium would theoretically occur when the H+ concentrations on both sides of the membrane were equal. However, because protons are constantly running through the system, it is nearly impossible for the system to reach complete equilibrium.
Having a hard time really getting it? Watch this Crash Course video on chemical equilibrium!
If you're still confused about diffusion itself, try watching this Khan Academy video!
